Permanent electrical stimulation of sacral nerve roots, sacral nerve stimulation (SNS), was first used in the late 1970s by Brindley et al. to treat neurogenic urinary retention (1). Just over ten years later, the first patient series showing positive results from the treatment of this patient group became available (1, 2). The first report on the effect of sacral nerve stimulation in three patients with faecal incontinence was published in 1995 (3).
In 1999, this method was introduced in the University Hospital of North Norway, the first Nordic hospital to do so. Sacral nerve stimulation is now used to treat a number of dysfunctions of the lower urinary tract, the pelvic floor and the bowel, and new indications are being tested. The method is minimally invasive and does not cause any structural changes in the pelvic floor. Even though sacral nerve stimulation is established throughout Norway today, it is our impression that the method is relatively little known in the primary health services, and not even very well known in the specialist health services.
Materials and methods
The article reviews sacral nerve stimulation and treatment results for various indications based on the authors’ own experiences of the method and a review of relevant literature retrieved by way of a non-systematic PubMed search. All the authors of this article perform sacral nerve stimulation at University Hospital of North Norway, and they also participate in the interdisciplinary working group at the Norwegian Continence and Pelvic Floor Centre.
Mechanism
Electrical stimulation is obtained by placing an electrode attached to a stimulator against one of the sacral nerve roots (fig. 1). The voltage used normally ranges from 0.5 to 3 volts, with a frequency of approximately 15 Hz. The effect was initially assumed to be caused by direct stimulation of the bladder muscle, the urethral sphincter and the anal sphincter via motor nerves (3, 4). Later studies, however, indicate that electrical stimulation of the sacral nerve roots causes complex modulation of motor, sensory and autonomous nerve paths in both the central and the peripheral nervous system (5) – (7). The complexity of this active mechanism is the most likely reason why the method can have an effect on several partly conflicting processes.
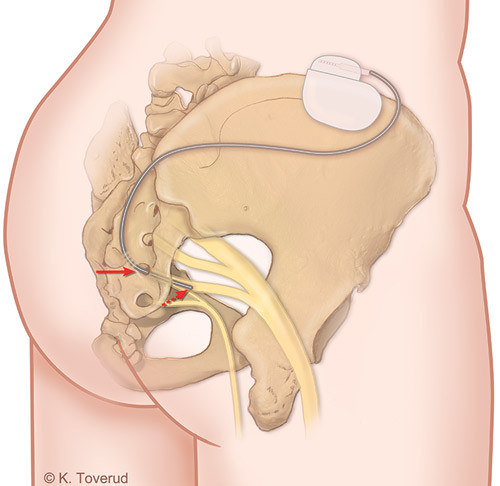
Surgical procedure
Currently, only one system for sacral nerve stimulation is commercially available (InterStim, Medtronic, Minneapolis, MS). The surgical procedures are identical irrespective of the indication in question. First, a peripheral nerve evaluation test (PNE test) is undertaken to establish whether the patient will benefit from sacral nerve stimulation. Under general anaesthesia, sedation or local anaesthesia one or several sacral foramina are cannulated, usually foramen 3 or foramen 4. When the cannula is inserted through the sacral foramen, the tip will be located immediately adjacent to the corresponding sacral nerve root (fig. 2).
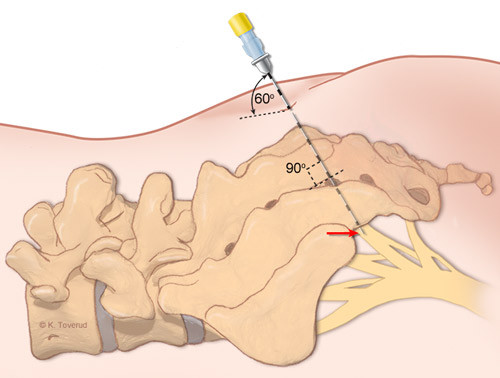
The cannula is supplied with pulsating current from a stimulator. If the operation takes place under general anaesthesia or sedation, the stimulation may lead to contraction of the anal sphincter (fig. 3). If it takes place under local anaesthesia, the stimulation may also cause a sensory response corresponding to the rectum, the bladder or the vagina. If an appropriate response is obtained, an electrode is inserted in the same place using percutaneous technique.
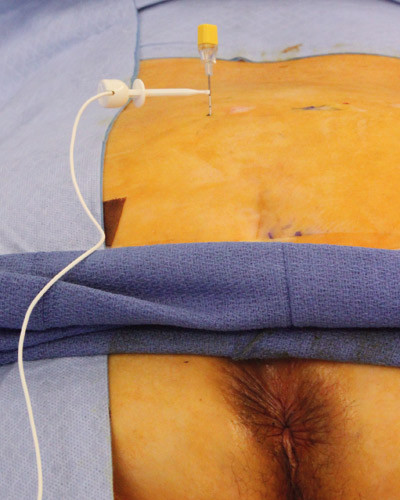
The electrode is linked to an external stimulator that the patient carries during the test period. The duration of this period depends on the patient’s condition. During the test period, the patient keeps a daily record of the frequency of the symptoms. If the test is carried out using a temporary electrode, this can be easily removed by the patient him/herself once the test period is over. If a permanent electrode has been used, the extension wire emerging through the skin at the flank can be cut at the skin level.
After completion of the test, the frequency of the symptoms prior to and during the test are compared. There is a broad agreement that a PNE test should lead to a reduction in symptoms of 50 % or more before implantation of a permanent stimulator is offered.
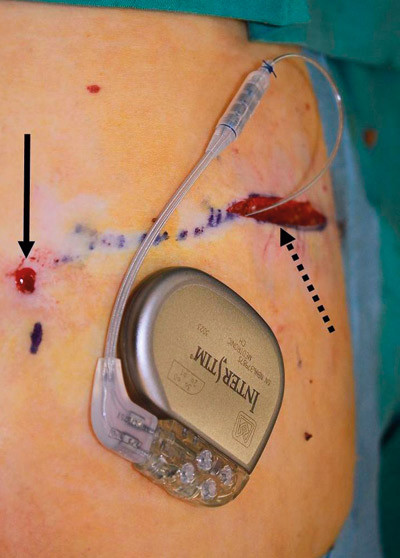
Two different stimulators are available – one with a service life of up to ten years and another, considerably smaller model, with a service life of 3 – 5 years. The stimulator is implanted deeply subcutaneously in the left or right buttock, according to the patient’s wishes (fig. 4). If the patient was tested using a temporary electrode, a permanent electrode is inserted during the implantation of the stimulator. If a permanent electrode was used for the test, the stimulator will be attached to this electrode. Patients are supplied with a remote control device that enables them to turn the stimulator on and off, change the voltage and switch between up to four different stimulation programmes.
Patient selection
Implantation must not be undertaken if there is a skin infection in the area, and a special evaluation must be made if the patient suffers from chronic inflammation of the skin (e.g. psoriasis) or has a weakened immune system. Care should also be taken with regard to persons with personality disorders, since they more often respond poorly in spite of a positive test period (8). In our experience, patients need to be highly motivated to undergo sacral nerve stimulation, and preferably be able to handle the remote control device that controls the stimulator. Furthermore, it is essential that the patients have realistic expectations regarding the results of the treatment, including the possibility of a technical failure and the need for stimulator reprogramming and possibly reoperation.
It has been assumed that persons who are likely to need an MR scan of the pelvic floor region later are not suitable subjects for sacral nerve stimulation. However, studies have failed to establish clearly any undesirable effects for the patients following MR scans (9, 10). This is also our own experience. However, a recent study reports that two patients had their stimulators damaged as a consequence of MR scanning of the pelvic region using a 3-tesla scanner (11). The necessity of an MR scan should therefore be balanced against the risk of damaging the stimulator.
Indications and results
Non-obstructive urinary retention
The first patients who were treated with sacral nerve stimulation suffered from neurogenic urinary retention (partial transverse lesion, multiple sclerosis and stroke) (1, 2). Sacral nerve stimulation can also be efficacious in cases of neurogenic urinary retention due to cauda equina syndrome, prolapse of the intervertebral disc, sequelae after spinal surgery and polyneuropathy (12). The treatment can also be efficacious in cases of idiopathic, non-obstructive urinary retention where other forms of treatment have failed (5, 12, 13).
A positive PNE test response is achieved in approximately 70 % of patients (14), after a test period of 3 – 7 days. Patients with a positive test response achieve a significant reduction of residual urine and use of a catheter once the stimulator is implanted (5, 13). Some studies also show a significant improvement in perceived health (5), healthrelated quality of life and depression score (15). Other studies have been unable to establish such correlations (13). A total of 50 %–80 % of those who have a stimulator implanted are experiencing a beneficial effect 3 – 6 years after the implantation (12) – (14).
Urinary urge incontinence
Urinary urge incontinence is characterized by bouts of a strong and irresistible urge to urinate without the bladder being full. The syndrome may also be accompanied by occasional incontinence (urge incontinence). The condition is initially treated with bladder training and antimuscarinic drugs (16). If this treatment has little effect, sacral nerve stimulation can be attempted.
Sacral nerve stimulation may also be efficacious, irrespective of whether any hyperactivity of the detrusor is detectable or not, in patients both with and without accompanying urinary incontinence (17). A positive PNE test response is achieved in 52 %–77 % of the patients (18) after a test period of 3 – 7 days. In patients who have a stimulator implanted, the effect is sustained in approximately 60 % after 5 – 6 years (12, 13).
Anal incontinence
Sacral nerve stimulation can be indicated in patients with faecal incontinence or gas incontinence combined with an inability to retain faeces when they experience an urge («urgency»), in cases where maximally conservative treatment has been ineffective. The PNE test is conducted over a period of 2 – 3 weeks. A positive test response is achieved for 70 %–90 % of patients (19) – (22). In those who have an implanted stimulator, the effect is sustained in 70 %–90 % after 2 – 5 years (19, 22, 23). Double-blind studies with the stimulator set so that patients were unable to determine whether it was turned on or off have shown that symptoms recur when the stimulator is turned off (24, 25).
The treatment has a positive cost-benefit effect, with a significant improvement in quality of life (26, 27). Patients with sphincter lesions covering up to one-third of the circumference can benefit from sacral nerve stimulation (20). However, sphincter lesions detected by ultrasound are a negative predictor of success (28), and large sphincter defects should probably first be treated by way of sphincter reconstruction.
Constipation
The documentation on the effects of sacral nerve stimulation on chronic constipation is more limited than for the conditions described above. Stimulation of the right or left third sacral nerve root has been shown to increase motility in the left colon and in the rectum (29). An effect has been found in patients suffering from intractable voiding dysfunction with normal and prolonged passage time (30, 31). The PNE test is undertaken over a period of 3 – 4 weeks. A positive PNE test response in cases of chronic constipation is achieved in 43 % – 73 % of patients (30, 32), and the effect is sustained in approximately 90 % after two years (30, 31). A double-blind study with the stimulator set so that patients were unable to determine whether it was turned on or off have shown that symptoms recur when the stimulator is turned off (33).
Other conditions
Sacral nerve stimulation may have an effect on chronic pelvic floor pains, for which all other available treatments have been attempted (34, 35). In a study of nine patients suffering from chronic pains in the anus or rectum, a positive PNE response was obtained for four patients. The median VAS pain score was reduced from 8 to 1, with a sustained effect for more than two years after the implantation of a permanent stimulator (36).
A pilot study has shown that sacral nerve stimulation can be efficacious in patients who are considerably distressed by irritable bowel syndrome (37), and a research group in Århus, Denmark, is currently investigating this in a prospective study.
Sacral nerve stimulation has also been attempted on patients who are at an acute stage after a transverse lesion. Bilateral permanent stimulation may also be efficacious in achieving voiding of the bladder and preventing development of neurogenic bladder dysfunction and motility dysfunction of the colon (38). If this is confirmed, sacral nerve stimulation may be implemented in the early treatment plan for patients with a transverse lesion.
Complications
After implantation, an infection develops in 2 % – 10 % of the patients (19, 39) and leads to removal of the implant in approximately half of the cases. Other complications include pain radiating to the perineum or a lower extremity, pain or discomfort around the stimulator or electrode, dislocation of the electrode with loss of efficacy, or loss of efficacy for no known reason (40, 41). However, this can often be corrected by reprogramming the stimulator, and up to 75 % of the patients will need this (42). Furthermore, 16 % – 54 % of patients will need reoperation, to replace the electrode after technical failure or to move the stimulator because of local discomfort (19, 31, 40, 41). A total of 8 % – 20 % of the patients will eventually have the equipment explanted because of technical failure, pain or discomfort (12, 19, 31, 43).
Follow-up
Since many of the patients will need reprogramming or reoperation, it is essential to have a good and readily available follow-up service (42). At the University Hospital of North Norway we have chosen to let the patient him/herself determine the need for follow-up. In addition, we have established an annual training course, including an offer for checking and possibly reprogramming the stimulator in connection with the course.
Conclusion
Sacral nerve stimulation can be an effective form of treatment of overactive bladder, non-obstructive urinary retention, faecal incontinence, constipation and chronic pain in the pelvic floor area. The treatment is minimally invasive, and the efficacy of the treatment can be determined by means of a brief test period. Most of those who have a stimulator implanted will retain the effect for a number of years. However, many patients will need reprogramming of the stimulator or reoperation to replace the electrode, and this necessitates close follow-up to maintain optimal efficacy.
- 2.
Tanagho EA, Schmidt RA, Orvis BR. Neural stimulation for control of voiding dysfunction: a preliminary report in 22 patients with serious neuropathic voiding disorders. J Urol 1989; 142: 340 – 5. [PubMed]
- 4.
Bazeed MA, Thüroff JW, Schmidt RA et al. Effect of chronic electrostimulation of the sacral roots on the striated urethral sphincter. J Urol 1982; 128: 1357 – 62. [PubMed]
- 10.
Uitti RJ, Tsuboi Y, Pooley RA et al. Magnetic resonance imaging and deep brain stimulation. Neurosurgery 2002; 51: 1423 – 8. [PubMed]
- 16.
Hunskår S. Behandling av overaktiv blære-syndrom. Tidsskr Nor Lægeforen 2005; 125: 2029-30. [PubMed]
- 17.
Groenendijk PM, Lycklama à Nyeholt AA, Heesakkers JP et al. Urodynamic evaluation of sacral neuromodulation for urge urinary incontinence. BJU Int 2008; 101: 325 – 9.
- 22.
Hollingshead JR, Dudding TC, Vaizey CJ. Sacral nerve stimulation for faecal incontinence: results from a single centre over a 10 year period. Colorectal Dis 2010; e-publisert 16.8.
- 41.
Datta SN, Chaliha C, Singh A et al. Sacral neurostimulation for urinary retention: 10-year experience from one UK centre. BJU Int 2008; 101: 192 – 6.
- 42.
Govaert B, Rietveld MP, van Gemert WG, Baeten CG. The role of reprogramming in sacral nerve modulation for faecal incontinence. Colorectal Dis 2011; 1: 78 – 81.